News and Blogs
Everything You Need To Know About Aerobic Culture Swabs
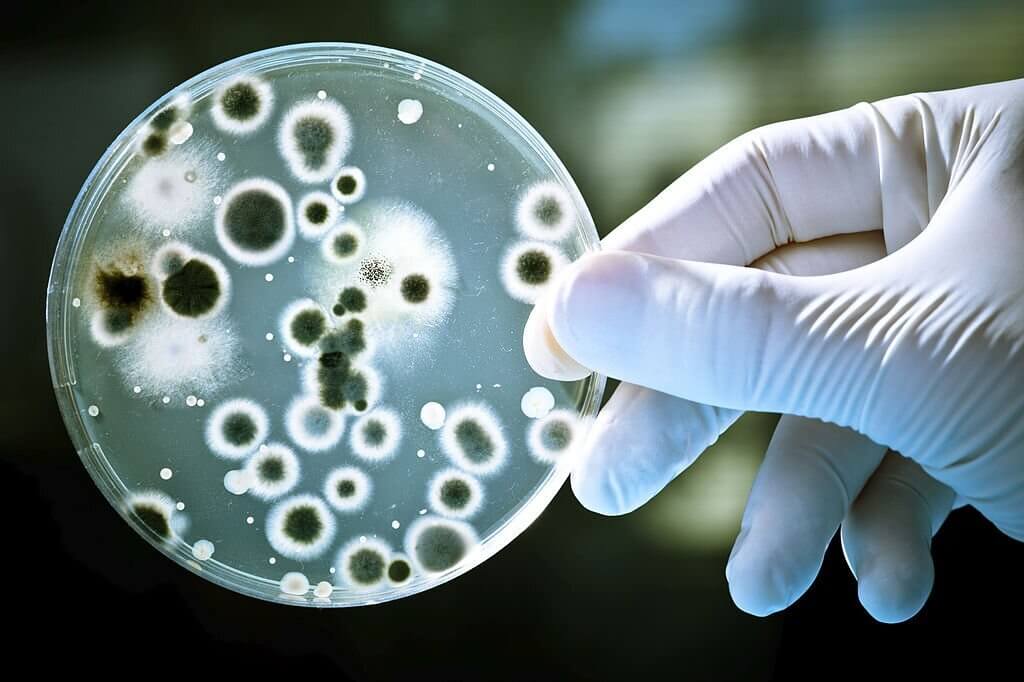
What are aerobic culture swabs?
Aerobic culture swabs are used to collect specimens from patients to test for the presence of aerobic bacteria. They consist of a swab tip made of synthetic material such as polyester, rayon, or nylon attached to a plastic shaft. The swab tip is sterile and is used to collect bacteria from the affected site on the patient, such as a wound, abscess, ulcer etc. The swab is then placed in a tube containing transport medium to keep the bacteria alive until it reaches the laboratory for testing. Transport mediums contain nutrients to facilitate bacterial growth and may also contain antibiotics to prevent overgrowth of commensal organisms. Common transport mediums used with aerobic culture swabs include Amies, Stuarts, Cary-Blair etc.
How are aerobic culture swab specimens collected?
Proper specimen collection technique is critical for accurate test results. The collection site should be cleaned to remove commensal flora and rinsed if any antiseptics were used. Depth of collection depends on the site; superficial swabbing should be avoided. Abscesses should be aspirated rather than swabbed. For wound swabs, tissue exudate or fluid should be targeted. Swabbing should be done by rotating the swab tip over a 1 cm area with sufficient pressure to collect adequate material. The swab tip should not be touched after specimen collection to avoid contamination. Once obtained, the specimen should immediately be placed in the transport medium tube and sent to the laboratory promptly, within hours if possible. Swabs allowed to dry out will not yield reliable results. Specimens should be stored and transported at room temperature, not refrigerated or frozen. Proper labeling with patient identifiers is critical.
How does aerobic culture swab testing work?
In the laboratory, the swab specimen is streaked onto agar plates, incubated overnight, and visually inspected for growth. Morphology of colonies and gram stain results are used to identify organisms. Biochemical testing may also be performed. Antibiotic susceptibility testing by methods such as Kirby-Bauer disc diffusion determines drug sensitivities. Automated systems like Vitek and MicroScan facilitate rapid identification and susceptibility testing. Positive cultures are worked up further if clinically significant. Culture can detect fastidious organisms like Haemophilus, Neisseria, streptococci etc. A disadvantage is that it may take 48-72 hours to finalize results while antimicrobial treatment decisions need to be made sooner. Anaerobic cultures require special handling and have lower yield from swabs. Still, aerobic culture remains the gold standard for diagnosis of most bacterial infections.
What are some limitations of aerobic culture swabs?
While extremely useful, aerobic culture swabs do have some limitations. Specimen collection quality is user dependent. Swabs only sample superficial material and may miss deeper pathogens, unlike tissue biopsies. Organisms present in low numbers may not grow. Fast growing species can overgrow slow growers. Prior antibiotic therapy reduces yield. Culture cannot detect viruses, which require molecular methods. Some bacteria like Mycobacterium tuberculosis require special media. Culture lacks ability to distinguish colonizers from pathogens without correlating to clinical findings. Contamination may lead to misleading results. Aerobic culture cannot detect resistance mechanisms mediated by genes not expressed in vitro. Overall, aerobic culture provides valuable data but results must be carefully interpreted in context of the patient and clinical scenario.
What are some guidelines for appropriate test ordering?
To maximize utility, aerobic culture swab testing should be ordered judiciously based on clinical judgement. Testing is indicated for suspected bacterial infections that are serious, lack obvious source, or do not respond to empirical treatment. Obtaining a culture prior to starting antibiotics provides a baseline record of organisms for comparison later if needed. For chronic wounds, repeat testing is appropriate if the patient fails to improve. Surveillance cultures are not necessary in the absence of clinical signs of infection. Once a pathogen's identification and sensitivities are established, repeat testing is unlikely to yield additional actionable information. Ordering cultures without a clear purpose wastes resources and may lead to misleading or uninterpretable findings without clinical correlation.
What are some future directions for aerobic culture techniques?
While traditional culture is still the mainstay of clinical microbiology, technological advances are improving speed and sensitivity of diagnostic testing. MALDI-TOF mass spectrometry enables rapid organism identification from culture colonies. PCR can detect fastidious bacteria directly from specimens within hours. Multiplex panels using syndromic molecular testing are emerging for different sample types. Next generation DNA sequencing enables culture-independent detection of all organisms, including those not culturable. As costs decrease and technology develops further, molecular techniques are expected to complement, though likely not fully replace, standard culture methods which continue to be the most cost-effective way to comprehensively evaluate for infection.
Click to View → Mantacc Aerobic Culture Swabs
Reference
Shah A, Ramola V, Nautiyal V. Aerobic microbiology and culture sensitivity of head and neck space infection of odontogenic origin. Natl J Maxillofac Surg. 2016 Jan-Jun;7(1):56-61. doi: 10.4103/0975-5950.196126. PMID: 28163480; PMCID: PMC5242076.
Related Posts
A Primer on Wound Culture Swab: Essential Facts & Guidelines






